Site brought to you by PolySciTech
Chapter 1
“Polymers are large macromolecules composed of many repeating subunits known as residues or monomers...” Professor Langweil droned on at the front of the 7:00AM UWHOC Introduction to Polymer Chemistry 305 class. He spoke in a continuous monotone voice pausing often just in case what he said became too interesting. In the back of the class, Steve could not take it anymore. Darkness overcame his eyes as his head drooped slowly forward pushing over his blue ink pen as it came to a rest on top of his textbook and half-scribbled notes.
Suddenly, a lithe hand grabbed his shoulder and shook it “Steve, wake up!” Jenny shouted. Steve snapped awake from his tumultuous dream to see the room sat empty save for himself and Jenny. Terror filled his mind as he looked up into Jenny’s calm face “Did you sleep the entire class?” She asked. He looked back down at his notes. He had scribbled the word ‘polymer’ and something about subunits before a long, blue line cut across the entire page where he should have written an entire class-worth’s of notes. “Oh, No!” he cried flipping the page over desperately hoping that somehow, in his dreams, he had taken accurate notes over the lecture on the other side. He looked up at Jenny and said “Yeah, I did. My room-mate stayed up all night playing PolySciTech Invaders and I couldn’t get any sleep.” He shook his head “I just can’t get polymers, like fundamentally, they don’t make sense.” Jenny sat down at the desk next to him and said, “Well, don’t try to think about polymers. Think about chains instead.”
“Huh?” asked Steve.
Jenny pulled out her notes and started to draw a sketch on the back of them. “Polymers are like chains. If you understand chains, you understand polymers.” Steve replied nervously “Jenny, you’re freaking me out.” Jenny shook her head and said “No, I’m helping you. If you pay attention, you might even pass this class.”
Reluctantly, Steve pulled out his notes. Jenny began:
“Just like chains, there are fundamentally two basic types of polymers. Crosslinked and uncross-linked. Think of what a typical uncross-linked chain looks like. It’s just a long, flexible whole with many links. Which looks like this:”

“Wow, you drew that incredibly well.” Said Steve. Jenny replied “Yes, I have talent in art too. Now, there is fundamentally, a different kind of chain which is crosslinked. You can imagine it with the chain segments all meeting together at a common junction and are bound to each other. Of course, it has to have more segments than two.”

“That’s incredible. Why does it have to be more than two, though?” Asked Steve as he scribbled a haphazard diagram of her picture. “Because, if it’s just two, it’s not a crosslink. It’s just another typical chain.”
Jenny continued “Now, I’ll explain how crosslinking is important. Picture if you will, a plastic chain floating just loosely in water. Can you see it?” Steve closed his eyes and imagined it, “Yeah, I guess if the plastic is light enough.” Jenny nodded and said “Let’s pretend it’s neutrally buoyant. This is an example of a polymer chain dissolved in a solvent. The chain flexes and wiggles around randomly. If, you had a whole bunch of chains kind of mashed together with no crosslinks, eventually they would just float away from each other. Can you see it?”
Steve pictured it and nodded “I can see it, but what does it have to do with polymers.” Jenny replied “ It’s a good model for polymer dissolution. A block of un-crosslinked polymer is just a bunch of polymer chains jammed together. When they dissolve in a solvent that has good attraction for the polymer chain, the polymer chains stretch out into a random-walk configuration. There’s some pretty complex math involved, but you can picture it just like the polymer chains floating around randomly in water.”
Steve nodded and replied “But what about your crosslinked polymer?” Jenny replied “Ah, good question. Picture this. It starts to float, but then the crosslinks hold it in place. No matter how much it wants to float away from the other polymer chains they are held in place and cannot move.” Steve said “So, then, what do crosslinked polymers do in solvent?” Jenny replied “They swell, but never dissolve. The amount of swelling is still related to how compatible the polymer chains are with the solvent, but no matter how good the solvent is, they will never dissolve.”
Steve furrowed his brows “So, they can never dissolve?” Jenny shook her head “Nope, not without destroying the polymer itself.”
Jenny drew a bunch of different chains at different lengths. She continued “Let’s talk a little bit about molecular weight. Polymers aren’t quite like small molecules, which have an exactly defined molecular weight based on their structure. In the case of polymers, molecular weight relates mostly to their chain-length. Think of a typical chain, the longer it is, the heavier it is. The correlation between the two is simple, it is the weight of a single link. The more links put together, the heavier the chain. It’s the same for a polymer. The molecular weight of a single residue, or monomer unit, times the number of residues in a row gives the molecular weight of that polymer chain. Due to their chemical formation, polymers are almost always a mixture of different lengths of chains. The word for this is ‘polydisperse’ which is to say they actually have many different molecular weights within the same polymer batch or set.” Steve frowned “So, how do you know their molecular weight at all?” Jenny replied “Well, you can’t ever have an exact molecular weight but you can determine an overall average molecular weight for the polymer chains as a group. There are a few different ways to calculate this, but that wasn’t part of this class.”
Steve shrugged and said “Ok. What about crosslinked polymers?”
Jenny replied “Determining molecular weight for a crosslinked polymer is a bit of a tricky issue, since the whole thing is technically ‘one-molecule’ as it is all crosslinked together. So, a 1 gram chunk of crosslinked polymer has a molecule weight of one gram. If you cut it in half, it now has a molecule weight of half a gram.”
Steve scribbled this down and looked oddly at his notes. Sometime during his nap he had half scribbled the word “biodegra…”
Memory flushed back to him. “Dr. Langweil also talked something about biodegradable polymers. What is that? Is it like magic or something?” asked Steve.
Jenny scoffed “Yeah, Steve, biodegradable polymers are made from glowing dust by pixies in toad-stool lined forests…” She looked and noticed he was writing this down. She grabbed hold of his pen to stop him “I’m being sarcastic!” She told him with a wry smile. She began again “There’s nothing magical about biodegradable polymers. Picture your chains again. Now, imagine one of the links breaking like this.

Steve marveled “Wow, you seriously have missed your calling.” Jenny said “No, I think polymer chemistry is right for me. Anyhow, when the link breaks, the main chain turns into two smaller chains.” Steve frowned “You mean; it doesn’t just disappear?” Jenny shook her head and said “Nope, it just gets shorter. This is called a chain scission, and can occur anywhere along the polymer chain. It can happen randomly on its own, or with the assistance of specific enzymes. If it happens a lot, you end up with a bunch of broken links floating around. Effectively, at that stage, the polymer has reverted to its monomers completely and is, effectively, gone.” Steve said “So, it just instantly disappears?” Jenny replied “No. Polymer degradation is a process. It takes time. The polymer chains just gradually lose weight with chain cleavage until they reach the final step. It is worth noting that with blocks of polymer, degradation can occur either primarily by surface erosion, affecting only chains exposed on the surface, or by bulk degradation of the chains inside the block as well.”
Steve asked “So, what could break one of these chains?” Jenny replied “Well, several things. For example, with polyesters, it is hydrolysis. Here water reacts with the ester unit to break it into an alcohol and an acid. ”
Jenny reviewed Steve’s notes “I think you were completely under for the part about making polymers. I’ll talk you through a few of these though.” Steve replied “So, is it just like bending links around a chain?” Jenny chuckled “Ummm… we’re going to set the chain metaphor down for this one. Think more about sophomore year organic chem.” Steve groaned “Ohhh… I know I memorized like two hundred reactions on flash cards for that but I totally can’t remember them all.” Jenny replied “That’s ok. We’re only going to talk about three basic ones. He said in class there were many others but we’re only doing three examples for now.”
Jenny pulled out her paper and sketched a traditional radical chain reaction.
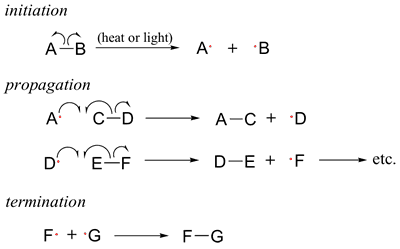
“Usually propagation is along a carbon-carbon double bond. Essentially vinyl-type materials participate in this reaction. It keeps propagating the free radical along the carbon-carbon double bonds until it terminates.” She said. Steven nodded vaguely remembering that part of organic chem so long ago.
Jenny continued “Of course, there are an incredible array of vinyl-type monomers which can be used for this. The professor said that a pretty cool use of these in the papers recently was to use certain types of monomers to create a reverse thermogel.” Steve asked “A what?” Jenny replied “A thermogel, it’s a polymer when dissolved in water makes it so that it is liquid at cold temperature and turns into a solid gel at higher temperature.” Steve shook his head “Wait, that’s backwards.” Jenny shrugged and replied “Yes, but recently it was used to make a gel which could safely kill anti-biotic resistant bacteria.”
Jenny continued “Not only the monomer type, but the initiation and termination steps are important too. Recently, people are finding new ways to initiate and control this reaction. You can even use another polymer to control this polymerization and end up with a graft polymer.”
Steve marveled. He said “Ok… I think I got a bit of it. What are the other two types?” Jenny flipped through her notes. “Another type is ring-opening polymerization. This is a kind where the monomer is a cyclic compound and it is split open by an initiator to propagate a chain. Typically, this only works under high heat with a catalyst. This is a popular one for making biodegradable polymers like poly(lactide) and PLGA.”
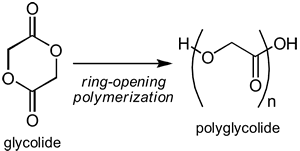
Steve noted it down and said “Have you ever noticed that when you talk, hyperlinks appear leading to more information?” Jenny rolled her eyes “Steve, that’s the weakest pick up line I’ve ever heard. Focus. There’s only one polymerization scheme left and it’s polycondensation.”
Steve marked it down “Condensing, like forming dew?” Jenny nodded “A bit, well, sometimes water is involved, sometimes. Polycondensation is where the monomers link together to form bonds. As part of this process, a molecule is emitted. For example, when acid-alcohols condense to form esters, water is emitted. An alternate example is the condensation reaction between diamines and dicarboxylic acids, a process used to make nylon.”
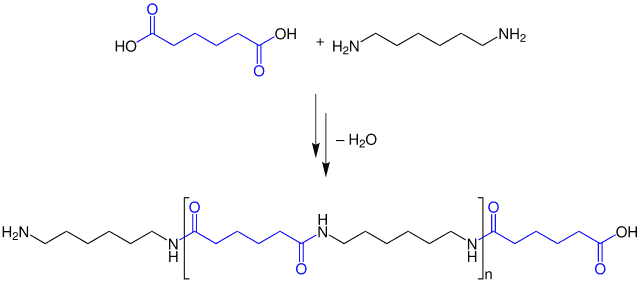
Steve scribbled this down. He asked “Is that all?”
Jenny replied “Of course not, there’s a bunch of others. There’s epoxy reactions which are typically based on conjugation reactions between carbon-oxygen based epoxy rings and either dialcohols or diamines. Another kind of reaction is polyurethane synthesis, which occurs by reacting isocyanates with alcohols or amines. This has been used to make degradable PEU’s by linking PLGA-diols together. In all cases, however, polymers have the same basic properties. They are linked chains of monomers.”
“Wow!” Steve said, closing his notes. “This is great to know. Hey, let’s say you wanted to buy polymers. Where would you go?” Jenny smiled and replied “PolySciTech... Where you can live a radical life by working with polymers.”